The bond angle between the two pairs bonded with the central atom is 180 degrees, which makes the molecular geometry of XeF2 linear.The central iodine atom in triiodide has sp3d hybridization.In triiodide anion, the central iodine atom has three equatorial lone pairs of electrons and the terminal iodines are bonded axially in a linear shape. The lone pairs are on the equatorial position to the bonded pairs. There are two pairs of bonded electrons and three lone pairs of electrons. Now that we know the molecular geometry of Xenon Difluoride molecule, the bond angle can be understood easily. XeF2 is a linear molecule due to the arrangement of fluorine atoms and the lone pairs of electrons in the symmetric arrangement. The shape of the molecule should be trigonal bipyramidal as per the hybridization, but it is not. These three lone pairs of electrons spread out in an arrangement that is on the equatorial position to the bonded pairs of electrons. These six electrons are now the non-bonding electrons. For Xenon, two electrons out of eight form bonds with the fluorine atoms. But here in XeF2, it is forming bonds with two Fluorine atoms only. This means that a single molecule of Xenon can form bonds with five molecules. Here the steric number for the central Xenon atom is 5. VSEPR is an abbreviation for Valence Shell Electron Pair repulsion theory. This theory is based on the steric number of the central atom and the valence electrons of the compound. The molecular geometry of Xenon Difluoride can be understood by knowing the VSEPR theory. But as Xenon does not form bonds easily, this compound is an exceptional case. Generally, the Lewis structure is helpful to understand the molecular geometry of any given chemical compound. Thus the hybridization of XeF2 molecule is sp3d. Hence the hybridization of the central atom Xe is sp3d. The arrangement of the electrons of Xenon changes to s2 p5 d1 with two unpaired electrons. Whereas in XeF2, the Xe molecule has an excited state. The ground state of the Xenon has 8 electrons arranged in s2 p6 orbitals. In XeF2, the outer shell of Xenon has eight electrons out of which two electrons participate in bond formation. During bond formation, two or more orbitals with different energy levels combine and make hybrid orbitals. Hybridization of a given molecule is vital to understand the geometry of the molecule. The total number of valence electrons for XeF2=22. A single molecule of Xenon has eight electrons, and a Fluorine molecule has seven valence electrons. So for this compound XeF2, there is one molecule of Xenon and two molecules of Fluorine. Whereas if there are less than eight electrons, the compound accepts the electrons from the other molecule to be stable. If there are more electrons than it, then that compound donates the electron. This rule states that every molecule should have eight electrons in its outer shell of an atom to be stable. Lewis structure is based on the octet rule. The bond formation in the compound are represented as straight lines and the lone pairs are depicted as dots. For distinguishing between the different types of electrons, both bonding and lone pairs of electrons are represented differently. The ones that do not participate in bond formation are called lone pair of electrons.

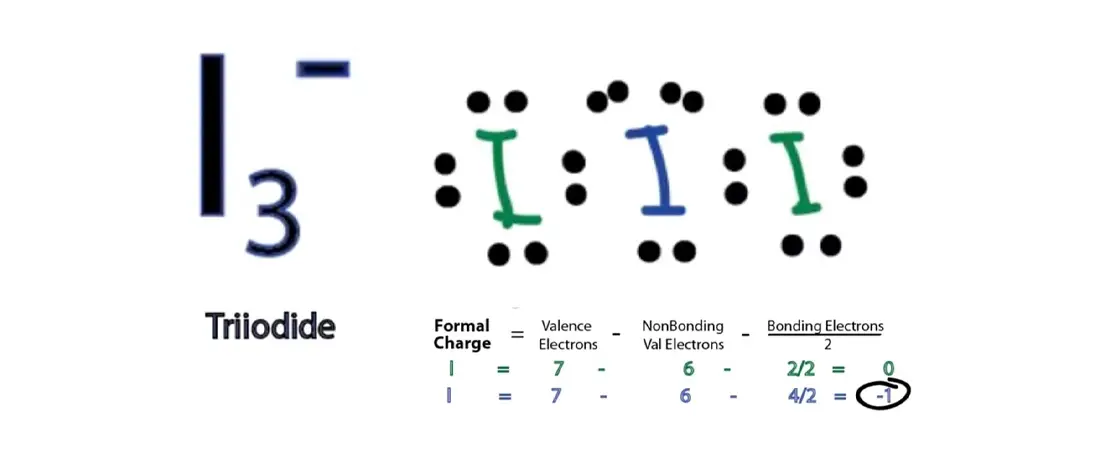
The electrons that participate in bond formation, as well as the ones that do not participate, are collectively known as valence electrons.Įlectrons that take part in forming bonds are called bonding pairs of electrons. This structure helps in understanding the charges on the molecules of the compound.
It is a pictorial representation of all the electrons participating in forming bonds. The Lewis structure of a given chemical compound is crucial for knowing all the physical properties and chemical properties.
